Link Your Antibody
to its Fullest
Potential
of services to include bioconjugation including antibody drug conjugation.
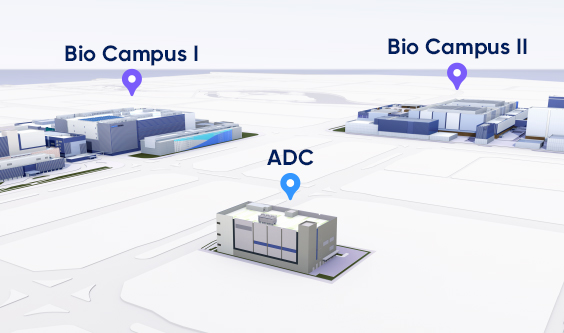
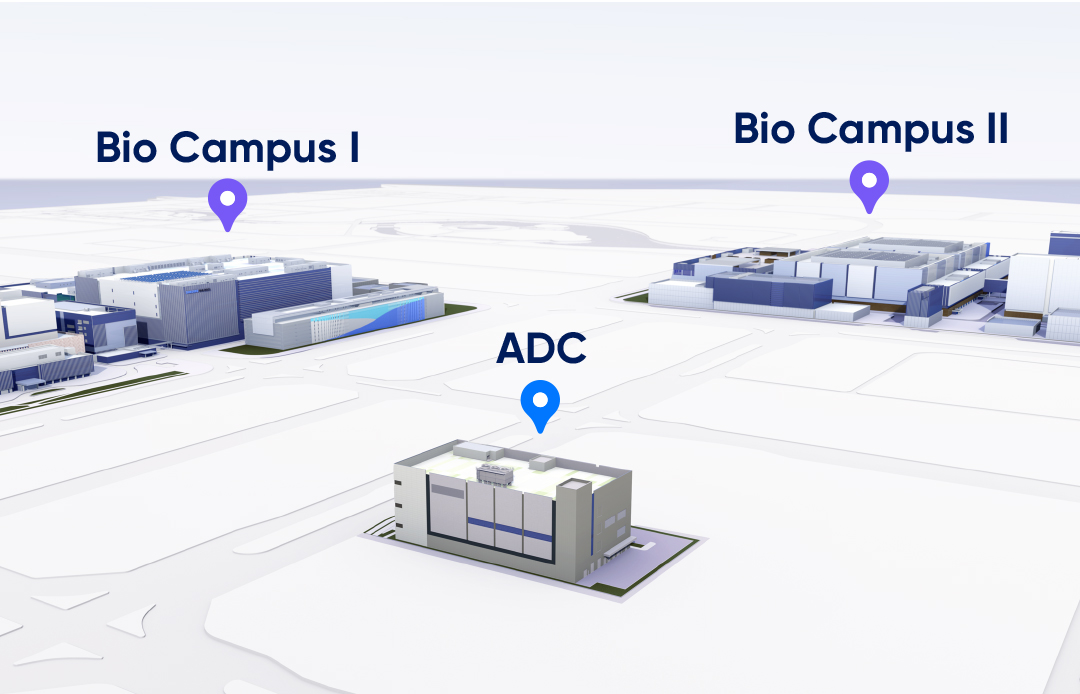
Expected to be operational in 2024, our standalone
bioconjugation facility will be located across from our main manufacturing
site and is supported by the robust GMP systems established for our existing
high-capacity clinical and commercial biologics manufacturing.
Dedicated Facility
-
- Expedited DS delivery with flexibility at a single site
- Expertise in quality and proven operational track record
- Integrated systems with established tools and procedures
- Stable supply chain management system
- Features
-
-
Single-use and stainless steel reactors
(Up to 500L conjugation train) - Full-scope laboratories for Development / QC / MSAT
- HPAPI facility
- Occupational Exposure Limit (OEL) target of 5ng/m³
-
Single-use and stainless steel reactors
Dependable ADC Service
We offer comprehensive services that range from antibody production and conjugation development to manufacturing.
Development Services
Expedite your timeline and development to clinical supply
-
Late discovery
Tool box options for the best development choice
-
Conjugation
Method, parameter optimization, DAR variation control
-
Analysis
ADC Method development, Precise Analysis and Characterization
-
Formulation
Stability enhancement and Lyo development
Concurrent development of mAb and ADC
- Cell Line Development
- mAb Process Development
- ADC Process Development
- Conjugation parameter development
- Downstream process development
- ADC Formulation development
- Analytical method development
- Non GMP mAb Tox, Non GMP ADC DS
- GMP mAb DS, GMP ADC DS
- 15 months
Manufacturing process
Maximum flexibility to accommodate various requirements
Payload & mAb Prep
- mAb Thawing
- Payload Prep
- UF/DF1
- Reduction
Conjugation
- Conjugation
- Quenching
- UF/DF2
- Chroma- tography
Formulated Bulk
- UF/DF3
- Formulation & Bulk Fill
Analytical Testing
Analytical methods to support ADC DS characterization and release of DS
-
Raw Material
- ID for payload/linker
-
IPC Testing
- Protein concentration
-
Chemistry
- DAR
- Residual solvent
- Free drug
-
Biochemistry
- Bioassay
- Antigen binding assay
- Cytotoxidity assay
Samsung Life Science Fund and securing future partnerships.
conjugation technology
development technology
technology
technology for ADC
